Category: Pharmacy - Page 2
Privacy and Security at Online Pharmacies: How to Protect Your Data in 2026
Learn how to protect your health data when using online pharmacies in 2026. Discover the only two trusted seals to look for, why most sites are unsafe, and what steps to take if your data is stolen.

WHO Model Formulary: International Standards for Essential Generics
The WHO Model Formulary sets global standards for essential generic medicines, ensuring affordable, safe, and effective treatments reach the people who need them most. Used by over 150 countries, it's the backbone of equitable health care.

Prescription Assistance Programs: Direct Help from Manufacturers
Manufacturer prescription assistance programs help millions afford costly medications through copay cards and free drug programs. Learn how they work, who qualifies, and the hidden traps that could leave you without coverage.

How Insurers Choose Which Generics to Cover: The Real Rules Behind Formulary Decisions
Insurers use strict criteria to pick which generic drugs to cover - focusing on cost, safety, and effectiveness. Learn how formularies work, why some generics are approved and others aren't, and what you can do if yours is denied.

Serious Adverse Events: How to Report Generic Drug Reactions Correctly
Learn how to properly report serious adverse events from generic drugs. Understand why these reactions are underreported, how to identify the manufacturer, and why your report matters for patient safety.

How to Verify Drug Authenticity: Official Tools and Resources You Can Use
Learn how to verify if your medication is real using official tools like the EU's FMD system, FDA resources, QR codes, and more. Protect yourself from dangerous counterfeit drugs.

The Science of Medication Safety: Understanding Risk, Benefit, and Real-World Evidence
Understanding how medication safety science identifies real-world drug risks beyond clinical trials, using data, statistics, and patient outcomes to protect lives and improve prescribing.

Quality concerns: when clinicians question generic manufacturing
Clinicians are raising alarms about the quality of generic drugs made overseas, citing higher rates of adverse events and unreliable manufacturing. Here's what's really happening behind the labels.

Verifying Your Prescription at the Pharmacy: A Simple Patient Checklist to Avoid Medication Errors
Learn how to verify your prescription at the pharmacy with a simple 6-step checklist to avoid dangerous medication errors. Know what to check, what to ask, and how to protect yourself.

Dangerous Medical Abbreviations That Cause Prescription Errors
Dangerous medical abbreviations like QD, U, and MS cause preventable errors that harm patients. Learn which ones to avoid, why they’re risky, and how to stop them from costing lives.
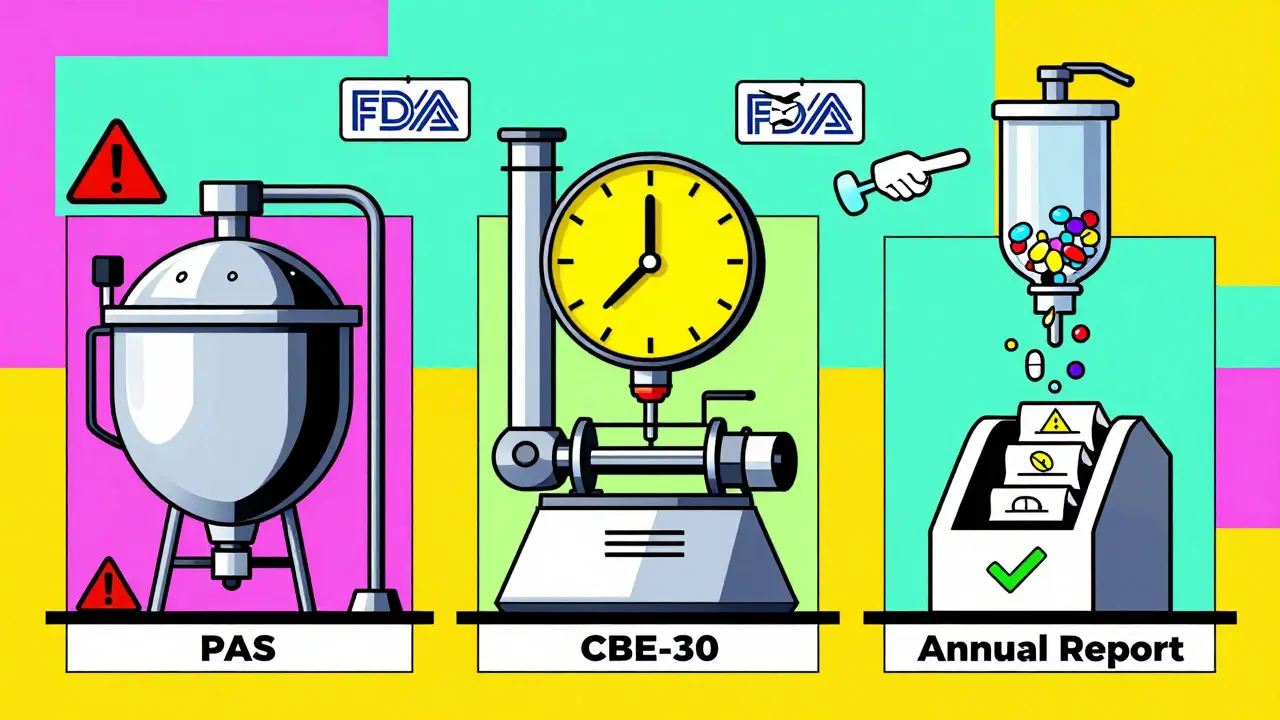
Manufacturing Changes: Notification and Approval Requirements for Pharmaceutical Quality
Understand the FDA's three-tier system for manufacturing changes in pharmaceuticals: major, moderate, and minor. Learn what triggers approval, notification, and annual reporting - and how to avoid costly regulatory mistakes.

Euglycemic DKA on SGLT2 Inhibitors: How to Recognize and Treat This Hidden Emergency
Euglycemic DKA is a dangerous form of diabetic ketoacidosis that occurs with normal blood sugar levels, often in patients taking SGLT2 inhibitors like Farxiga or Jardiance. It's easily missed but can be fatal without prompt recognition and treatment.