What Are Authorized Generics, Really?
An authorized generic isn’t a knockoff. It’s the exact same drug as the brand-name version-same active ingredient, same factory, same packaging-but sold under a generic label and usually at a lower price. The brand-name company makes it themselves and sells it to pharmacies as a generic. No ANDA filing. No third-party manufacturer. Just the original maker cutting into their own profits to beat competitors at their own game.
This isn’t some loophole. It’s a legal, FDA-approved strategy that’s been around since the 1980s, after the Hatch-Waxman Act changed how generics enter the market. The FDA has tracked every single authorized generic since 1999, and between 2010 and 2019, there were 854 of them launched. That’s not noise. That’s a pattern.
Why Do Brand Companies Use Them?
It’s simple: control. When a patent expires, any generic company can step in and undercut the brand. But if the brand company launches its own generic version right away, they can grab 30%, 50%, even 70% of the market before the real generics even get off the ground.
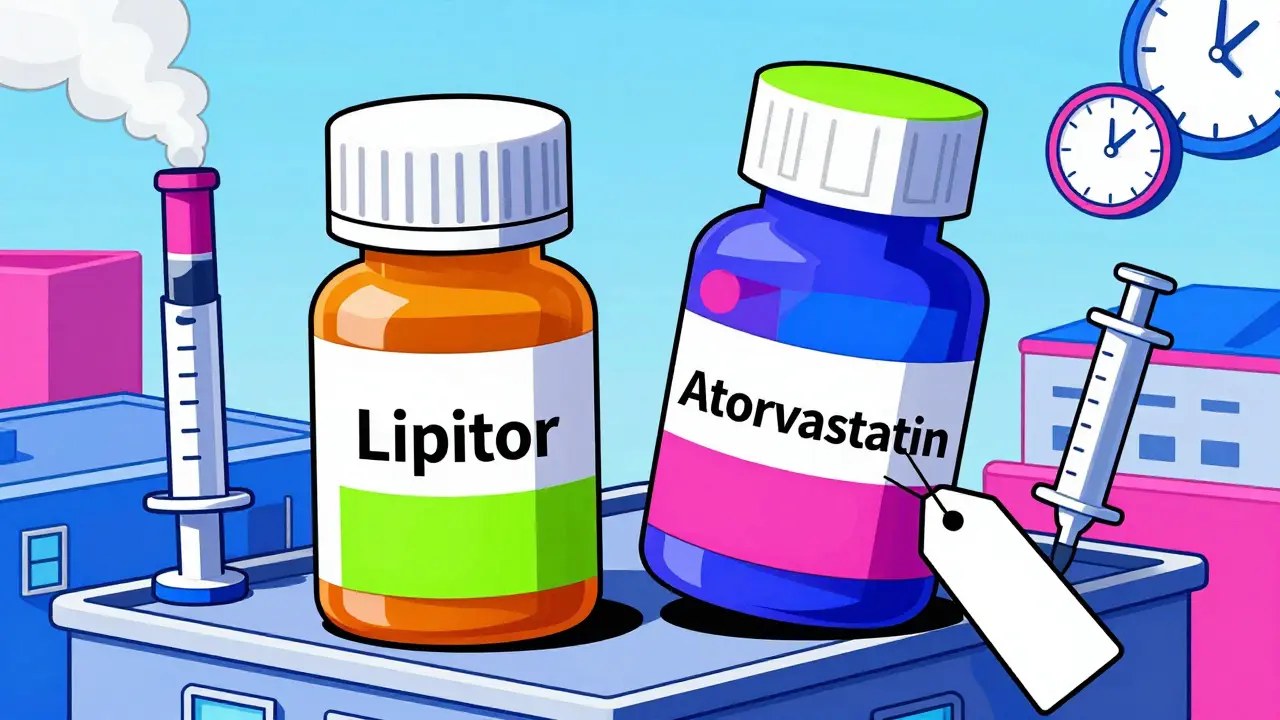
Here’s how it works in practice. Let’s say a drug like Lipitor loses patent protection. Instead of letting a competitor like Teva or Mylan swoop in with a cheaper version, Pfizer might launch its own authorized generic the same day. It’s the same pill, same bottle, same dosage-but now it’s labeled as "atorvastatin" and priced 40% lower than the brand. The brand keeps the revenue stream, the pharmacy gets a cheaper option, and the real generics? They’re stuck with the leftovers.
And here’s the twist: most authorized generics don’t launch until after the first traditional generic gets approved. Why? Because the brand doesn’t want to cannibalize its own sales too early. They wait until the market is ready to shift. Then they drop the hammer.
Where Are They Most Common?
Not all drugs get authorized generics. It’s mostly oral tablets and capsules-things that are easy to replicate. You won’t see many authorized generics for injectables or inhalers. Why? Because those are harder and more expensive to make. The cost of setting up production for an authorized generic only makes sense if the drug sells in massive volumes.
That’s why you’ll see them in high-volume markets: blood pressure meds, cholesterol drugs, diabetes pills, antidepressants. These are the drugs that make billions for brand companies. When their patents expire, the stakes are high. That’s when the authorized generic comes out.
Between 2010 and 2019, three-quarters of authorized generics launched after the first traditional generic got approval. That’s not random. That’s strategy. They’re not trying to replace their own brand-they’re trying to crush the competition.

What’s Changing in the Market?
Here’s the big shift: the delay tactic is fading.
Back in 2014, authorized generic launches peaked. Companies were hoarding them, waiting months or even years to drop them in, just to squeeze every last dollar out of the brand. But that’s changing. According to RAPS in June 2025, fewer companies are holding back. Why? Regulatory pressure. Public scrutiny. And the fact that the market is just moving too fast.
Now, in markets where a generic company gets 180 days of exclusivity, about 70% of authorized generics launch before or during that window. That’s not a coincidence. It’s a direct response. The brand companies realized: if you wait, you lose. So they’re launching earlier, faster, and more aggressively.
And it’s working. The U.S. generic drug market is expected to hit $196.9 billion by 2034. That’s up from $138.2 billion in 2024. Why? Because over the next five years, $217 billion to $236 billion worth of branded drugs will lose patent protection. That’s a tidal wave of opportunity-and authorized generics are right in the middle of it.
The FDA’s New Move: U.S. Manufacturing Priority
In October 2025, the FDA announced a pilot program that’s going to shake things up. They’re now prioritizing ANDA reviews for generic drugs made and tested entirely in the United States.
This is huge. For years, most generic drugs-authorized or not-were made overseas. India, China, the Philippines. But now, the FDA is saying: if you want faster approval, build it here. That’s going to push brand companies to make their authorized generics in U.S. facilities, even if it costs more.
Why? Because speed matters. The first generic to market gets the biggest slice of the pie. If you can shave 30 days off your approval time by manufacturing locally, you win. And that’s exactly what the FDA is betting on.
This change could mean fewer authorized generics made overseas. It could mean higher prices for a while, as companies adjust. But long-term? It could make the supply chain more secure-and maybe even more transparent.

What About Biosimilars?
Authorized generics have mostly been about pills. But now, biologics are coming off patent. Drugs like ustekinumab and vedolizumab-used for psoriasis, Crohn’s, and other autoimmune diseases-are losing exclusivity in 2025 and beyond.
These aren’t simple pills. They’re complex proteins made in living cells. You can’t just copy them. That’s where biosimilars come in. And yes, brand companies are already thinking about launching authorized biosimilars.
The opportunity? Up to $25 billion in oncology and immunology markets by 2029. That’s a massive prize. And if the same playbook applies-brand makes it, sells it as a biosimilar, beats the competition-then we’re about to see a whole new wave of authorized generics. Only this time, they’ll be injectables, not tablets.
Are Authorized Generics Good or Bad for Patients?
It’s complicated.
On one hand, they bring down prices. They give patients cheaper access to the same medicine. In 2024 alone, generics and biosimilars saved the U.S. healthcare system $467 billion. That’s real money.
But here’s the catch: they’re not always about saving money. Sometimes, they’re about stopping competition. When a brand company launches an authorized generic right before a cheaper competitor enters, they’re not lowering prices-they’re locking out the real bargains.
A 2025 study in JAMA Health Forum found that when brand companies stretch out exclusivity, it costs commercial insurers $2.5 billion and Medicare $2.4 billion over three years. Drugs like imatinib and celecoxib are prime examples. Authorized generics can be part of that game.
So are they good? Sometimes. Are they fair? Not always. They’re a tool. And like any tool, they can be used to help-or to control.
What’s Next?
The future of authorized generics isn’t about more of them. It’s about smarter ones.
With more drugs going off patent, and more pressure on pricing, brand companies can’t afford to wait. They’ll keep using authorized generics-but faster, earlier, and more often. The days of dragging their feet are over.
And with the FDA pushing for U.S.-made drugs, we’ll see more authorized generics produced locally. That could mean higher costs at first, but better supply chains and fewer shortages down the road.
The real winners? Patients who get access to cheaper versions of the drugs they need. The real losers? Companies that think they can game the system by holding back competition.
The market is changing. And authorized generics are no longer just a tactic. They’re becoming a standard part of how drugs are sold after patent expiration. Whether that’s good or bad depends on who’s holding the reins-and who’s paying the bill.



Roshan Gudhe
February 5, 2026 AT 02:26Rachel Kipps
February 5, 2026 AT 05:08Alex LaVey
February 5, 2026 AT 18:06Jhoantan Moreira
February 7, 2026 AT 06:23Justin Fauth
February 7, 2026 AT 23:32Demetria Morris
February 8, 2026 AT 03:23Susheel Sharma
February 9, 2026 AT 20:18Janice Williams
February 10, 2026 AT 04:36Ed Mackey
February 11, 2026 AT 06:58caroline hernandez
February 12, 2026 AT 05:49Meenal Khurana
February 13, 2026 AT 19:51Sherman Lee
February 14, 2026 AT 05:53Keith Harris
February 15, 2026 AT 12:28Kunal Kaushik
February 16, 2026 AT 18:34